Reliable and rapid OneStep detection of toxigenic C. difficile without re-testing. Greiner Bio-One adds another CE-IVD certified molecular test to the Genspeed® test portfolio for nosocomial infections.
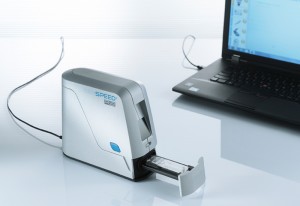
The portfolio of CE-IVD certified molecular diagnostic tests of Greiner Bio-One for the detection of nosocomial infections is growing. The Genspeed® C.diff OneStep test identifies toxigenic C. difficile by combining detection of Glutamatdehydrogenase (GDH), Toxin A, Toxin B and binary toxin in a single, molecular diagnostic test. The complete analysis including the detection on the Genspeed® R2 Analyzer takes under 100 min*.
The new test addresses one of the most serious threats to health care systems worldwide, the infection with Clostridium difficile infections (CDI), which is believed to be the most common healthcare-associated infection. The disease is causing antibiotic-associated diarrhoea (AAD) that may lead to pseudomembranous colitis and even to death1. In a 2013 published report, the Center for Disease Control (CDC) in the USA categorized C. difficile infections as “Threat Level Urgent”, the highest level available2.
OneStep ahead – a new way to detect C. difficile
The Genspeed® C.diff OneStep test features a simple, one-step molecular diagnostic test following the motto “OneStep – one test – four results”. The test is avoiding the currently used sequential 2-step diagnostic test procedures, which combine different test systems and assay principles for GDH and the C. difficile toxins. The rapid Genspeed® C.diff OneStep test pro-vides conclusive results without the need for confirmatory re-testing and enables inter-laboratory comparisons of test results.
Ready-to-use reagents and automated dispensing minimize the number of manual process steps within the workflow. The Genspeed® R2 Analyzer represents a reliable solution for customers interested in fast and accurate results. The virtually service-free system is simple to operate and is available at an attractive cost-benefit ratio.


Literature:
1) European Centre for Disease Prevention and Control. Point prevalence survey of healthcare-associated infections and antimicrobial use in European acute care hospitals. Stockholm: ECDC; 2013
2) Antibiotic Resistance Threats in the United States, 2013; www.cdc.gov/drugresistance/threat-report-2013 , U.S. Department of Health and Human Services, Centers for Disease Control and Prevention
(*) Time can vary with validated PCR-cycler used
You can order from us now under the following item numbers:
| Item N° | Description | Packaging |
|---|---|---|
| 453227 | Genspeed® Starter Package R2 (Genspeed® R2, PCR-Cycler, Notebook, Accessories) | 1 pc. |
| 453224 | Genspeed® R2 Analyzer | 1 pc. |
| 453228 | Genspeed® C.diff OneStep Test Kit (incl. Preanalytics Kit) | 48 Reactions |
| 453229 | Genspeed® C.diff OneStep Test Kit (incl. Preanalytics Kit) | 16 Reactions |
 Terugblik LabAutomation event 2019
Terugblik LabAutomation event 2019 Telkens u een pipet neemt, houdt u de data-integriteit in een hand.
Telkens u een pipet neemt, houdt u de data-integriteit in een hand. ELISA Methods with Biomek Workstations
ELISA Methods with Biomek Workstations Biomek Genomic Workstations
Biomek Genomic Workstations



 en (een) partner voor de lange termijn voor de farmaceutische industrie, biotechnologie, diagnostiek en de medische technologie.
en (een) partner voor de lange termijn voor de farmaceutische industrie, biotechnologie, diagnostiek en de medische technologie.